Note: In this blog I’m only going to discuss tetany in the context of breathwork since people are often interested in why such a physical change occurs. I’m not going to touch on other medical reasons that can cause tetany in other ways.
In the context of breathwork, tetany or “the breathwork claw” is a side effect that causes the fingers of the hand to typically close inwards into a lobster claw shape.
What it typically looks like when you are doing breathwork is below.

This is a temporary reaction by the body to what is going on at a chemical level in the body/blood.
So, why does this happen?
When we do Conscious Connected Breathwork, we are temporarily, deliberately changing the way we breathe, removing the pauses/gaps in our breath pattern. This usually means that we end up breathing out more gas than normal. Since the main “waste” gas we breathe out is Carbon Dioxide (CO2), then we expel more CO2 than normal. This loss of CO2 is more pronounced if we are breathing very fast or hard, especially when using too much force or effort on the out breath. CCB facilitators are very often heard saying things like “a soft outbreath”, or “let the out-breath fall out of your body”, “don’t push the out-breath”. Often this level of softness can be challenging since it requires us to “let go” of the out breath, and thus also let go a level of emotional control.
If we breathe out more CO2 than our body is generating (CO2 is a biproduct of processing glucose), then this changes the pH level of our blood (pH is the scale of how acidic to alkaline a liquid is).
Since CO2 is a kind of gas-version of acid (mixed with water it makes a weak acid called carbonic acid), then we are effectively removing acid from our system faster than we are adding it, thus we end up more alkaline.
Our body is very keen on our blood staying at just the right pH level, not too acid and not too alkaline (typically at 7.4). The normal resting range is 7.35 to 7.45, but if you run a marathon you might get down to 7.1 and during breathwork you might get up to 7.6. Our bodies are able to adapt to these temporary shifts which are in fact stress-testing the system. [Note that the pH scale is from 1 (very acidic) to 14 (very alkaline) with 7 being perfectly neutral, so we see that the body prefers the blood to always be very slightly alkaline.]
The body has a number of ways to affect the pH which work at varying speeds:
1. The Chemical buffers – acts in milliseconds
These are the bloods internal “shock absorbers”, normally used to remove small amounts of acid from the blood, like lactic acid from sprinting for the bus.
These are limited in capacity and can easily be overwhelmed if you keep breathing off CO2.
2. The Breathing Override – acts in seconds
This could be an apnea, where the brain stops the breathing process completely (holding your breath) to retain CO2 and allow more of it to build up internally, increasing the CO2 level.
3. The Protein-Calcium lock – seconds or minutes
This is what generally causes tetany and what we will be mostly looking into.
4. The Renal Clean-up – hours or days
If the blood pH level remains away from the expected level, then eventually the kidneys start to adjust how much bicarbonate (anti-acid) they are pushing into the blood stream, the kidneys can either increase or decrease the level depending on what is going on over time but generally the body uses this to make the system less acidic. When a client does CCB then the kidneys might adjust their levels which is why it is always recommended to drink plenty of liquids afterwards, including the day after too.
Looking at the Protein-Calcium lock in detail
When the blood becomes more alkaline (less acid) then the body starts looking for “acid” to bring the pH back to a more neutral level.
But before we look at this in detail, I realised that I wanted to really understand what “acid” meant. If you already know then you can skip the next section.
The Chemistry behind Acidity
In this case “acid” is effectively having lots of Hydrogen ions (H+) in the blood/plasma, which is mostly water, and water is probably not as simple as you might think.
I always thought of water as just H2O. Everyone’s heard of that name and it sounds nice and simple. But under the surface, water is constantly shifting between H2O and a combination of H3O+ and OH– (which when recombined make two lots of H2O). This is called auto-ionization. Here we can think of the H3O+ as H2O plus an H+ (acid) and the OH– as H2O with a missing H+ (alkaline).
H2O + H2O <=> H3O+ + OH–
(which is equivalent to)
H+ + H2O + OH–
When we have pH neutral water then the number of H+ and OH– floating around, changing back and forth are basically equal. This is why water is such a great base for acids, because it already has the ability to include H+ ions.
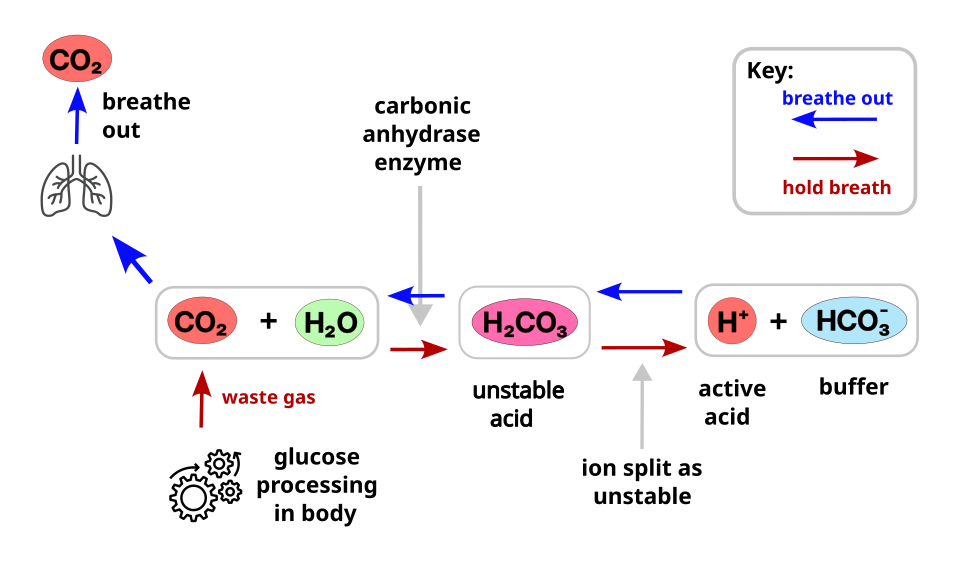
If we add CO2 (carbon dioxide) to water in the body, an enzyme in the blood helps them combine into a very unstable form of acid called Carbonic Acid (H2CO3), and this rapidly breaks down into H+ and HCO3–. Thus we end up with extra H+ in the water (higher ion density), and some dissolved Bicarbonate ions (the HCO3–).
If we remove CO2 by breathing out, then basically the opposite happens. An existing HCO3– grabs a spare H+ and turns back into H2O and CO2, and the CO2 is then expelled from the system.
The interesting thing in the body though, is that CO2 is constantly being produced as a waste product as we process glucose into energy. There is a constant addition of acid into the blood. The exit point for this CO2 is breathing out. If we stop breathing out then the CO2 / acid / H+ in the blood gradually rises. In a normal resting state the amount of CO2 the body produces should match the amount of CO2 we are breathing out. This then controls the number of H+ in our blood.
A Hydrogen ion (H+) is actually a proton that really wants an electron to be orbiting it, but hasn’t got that electron. Each H+ is like a window frame that needs glass in it, much more strongly than other items containing glass need their glass, such that the window frame will take glass from other structures that it gets close to, breaking the other structures to get their glass.
This is how acid “burns” through substances like metal. For example, if we take a neutral metal like Aluminium and add Hydrochloric Acid, then each H+ in the acid has a stronger “need” for the electrons (the “glass”) in the Aluminium than the metal itself – thus you get a fizzing effect as the H+ steal electrons to turn into a stable H2 hydrogen gas (the hydrogen has regained its natural number of electrons) and the Aluminium ends up as a water soluble version of itself (Al3+), which then dissolves into the water part of the acid. Note that you cannot really have “pure” acid, just very acidic water (or other base) which contains a lot of H+ compared to normal. Also, this kind of aggressive acidic behavior is very much not happening in our blood, but it demonstrates the chemistry.
So, having overly acidic blood (lots of wandering H+) is not something your body is happy about since the H+ tends to go around changing and breaking things. Not what we want inside our body, even if it’s only happening a little.
To manage this situation the body has an active defense running all the time to soak up spare H+ ions. This is a baseline level of bicarbonate the body keeps in the blood, which as we’ve seen before is HCO3–. This will bond to spare H+ to keep the number of H+ floating around under control. Ideally there is a ratio of something like 20:1 of HCO3– : H+ in the blood which produces a pH of roughly 7.4. We need more HCO3– since the H+ is aggressively looking for something to bond to, so having plenty of spare capacity saves the H+ finding another source the body was not expecting.
Ideally there is a ratio of something like 1:20 of Acid (CO2) : Alkali (HCO3–) in the blood which produces a pH of roughly 7.4, slightly alkaline. The body keeps 20x more HCO3– since the acid (H+ ions) is aggressively looking for something to bond to, so having plenty of spare capacity saves the H+ finding another source the body was not expecting.
Interestingly, the acid strength (concentration of H+ ions) that this ratio produces can be rapidly altered when CO2 is added or removed. If we look at the result of adding a few extra CO2 (thus H+ and HCO3– ions) we see that the acid concentration can shift very fast.
| Action | Dissolved CO2 : Bicarbonate | Acid Concentration | Notes |
| Baseline | 1 : 20 | 0.05 | Normal concentration |
| Add 4 | 5 : 24 | 0.2 | Adding 4 produces a 4x change in concentration, a 400% increase |
| Add 10 | 11 : 30 | 0.36 | Adding 10 produces a 7x change in concentration, 700% increase |
| Remove 0.5 | 0.5 : 19.5 | 0.025 | A change down by 50% |
This shows how quickly we can impact the concentration of H+ ions in the blood with small changes to the amount of CO2 in the system due to the baseline ratio difference.
When we connect the breath, continually breathing out CO2, the blood quickly ends up with too few H+ in it, which the body (and chemistry) is also trying to avoid, which brings us back to…
Back to the Protein-Calcium lock
When there is a lack of H+ in the blood due to breathing out lots of CO2 then this affects a protein/compound in the blood called Albumin. This is a complex molecule which normally has lots of negative slots in it that H+ likes to bond to (negative and positive bind together like magnets).
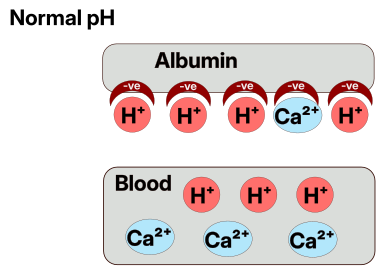
However, when the blood needs more H+ since there is not enough, then the Albumin gives up some of its own H+ into the blood to balance the pH. However, this leaves a negative space in the Albumin, acting kind of like a specialized chemical vacuum that attracts positively charged ions. Another type of ion that is floating around in our system are Calcium ions. If these get the chance they will bond into the negative spaces in the Albumin instead of being where they are supposed to be.
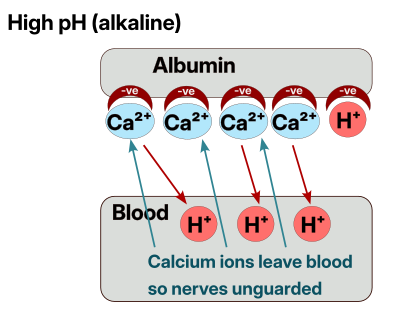
For completeness, if the blood is too acidic (you are holding your breath or your breath is very shallow) then we end up with too many Calcium ions being released into the blood, see later for the effects this has.
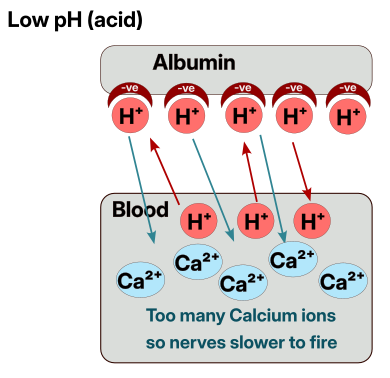
The cause of tetany
Nerves fire when Sodium (Na+) enters a nerve trigger gate. A gate can be opened by a “signal” like physical touch for a sensory nerve, or chemically for other nerves. When the gate is opened then Sodium can enter and fire the nerve. The thing that is “guarding” this gate are Calcium ions (Ca2+) which increase the electrical threshold required for the gate to accept triggering Sodium ions. If we remove the Calcium ions then much lower electrical signals (the body is full of small signals) will trigger nerves to fire as the Calcium ions are no longer guarding the gate.
This means that nerves are firing more easily than normal since the sensitivity of the nerves is not being dampened by the Calcium ions that are now bound to the Albumin instead of doing their normal job.
As all the nerves of the hand become more sensitive, this initially causes tingling sensations, and as the sensitivity increases can eventually cause the muscles to twitch or fire/tense up. Since most peoples muscles for gripping things are stronger than the ones for opening your hand, if all the muscles in the hand become tense, then the hand will close up and you will not be able to move it until the Calcium ions are released back into the blood to continue their normal role.
When we stop exhaling the CO2 so fast, the CO2 and thus H+ builds up to normal levels in the blood very quickly and the Calcium ions are then released back into the blood and everything goes back to normal – the nerves stop firing so easily and the muscles can relax.
Why is shifting to a more alkaline state a useful cleanout?
Everything in the body is always trying to find balance, there is no ideal, just an equilibrium. When there is a lot of H+ (acid) in the blood, then most/all of the Calcium ions are kicked off the Albumin into the blood, and act as surplus “guards” for the nerves. Thus the nerves become sluggish to fire and the body and sensations feel slow and dull.
When we are living in chronic stress, often holding our breath due to situations or tension, the CO2 builds and so does the acid in our blood.
Acidic blood => High Free Calcium => Numb Nerves
Thus stress is not only affecting your mood, it is physically affecting your ability to feel by dampening your nerves.
During breathwork we deliberately reduce the H+ in the blood and reduce the number of Calcium ions guarding the nerves.
Tetany is an indication that we are pushing too hard, we have stepped slightly over our own current window of tolerance to blowing out CO2. In CCB we don’t need to try so hard, even with a small shift in blood pH (minor tingling) we are getting all the benefits of waking up the nerves.
This increased nerve response helps the brain to recalibrate these nerves, or possibly to actually notice for the first time in a while that these nerves are still present and active – it wakes us up.
We literally regain the ability to fully feel our bodies again.
And once we can feel it, we can bring awareness, change and more life into our bodies.
Wrapping up
Understanding the body chemistry of “The Claw” highlights another aspect of how Conscious Connected Breathwork can move us from a “Grey” life into a more vibrant one, even on a purely physical level.
Whether you’ve spent decades overclocking your mind while your body suffered in silence, or have reached a point where it feels like no amount of talking can shift you out of feeling numb, maybe it is time to work directly with your body using the power of your breath.
If you are interested to experience this breathwork yourself, I am offering one-to-one sessions in North London, EN4.
Maybe we can breathe together soon.
And finally, how can we have a blog about “The Claw” without referencing this Jim Carey movie (final scene…)

Link: YouTube
Leave a Reply